the periodic table families
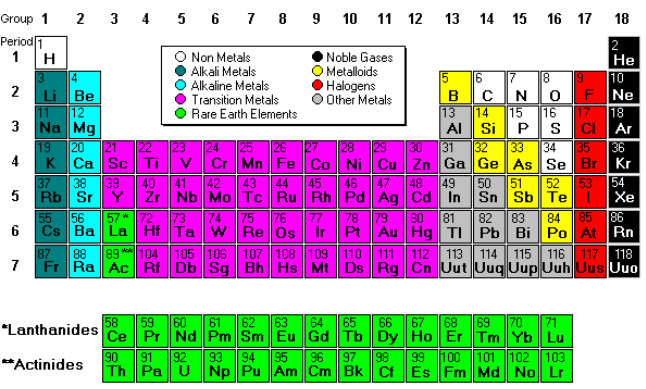
This Periodic Table shows that there is nine different families some example are the Non metals and Alkali Metals and so forth down the line, as seen on the image to the side.
The Periodic table can be divided into nine families of elements each having similar properties. The families include:
Alkali Metals
Group 1 of the periodic table are the alkali metals. They are highly reactive and do not occur freely in nature. They have just one electron in their outer shell, and they are ready to lose it in ionic bonding with other elements. They are malleable, ductile, are good conductors of heat and electricity and are softer than most metals.
Alkaline Metals
Group 2 includes the alkaline earth elements. They are metallic elements and have an oxidation number of +2, making them very reactive.
The Transition Metals
Groups 3 through 12 include 38 elements called transition metals. They are malleable and ductile and also conduct heat and electricity. They have valence electrons in more than one shell and exhibit several common oxidation states.
Other Metals
Groups 13, 14, & 15 include the other metals elements. They are malleable and ductile but are not the same as transition elements. They do not exhibit a variety of oxidation states, they have valence electrons only in the outer shells. They are solid, have high density, and are opaque. Their oxidation numbers are +3, +/14, and -3.
Metalloids
Metalloids are found between the metals and non-metals along a boundary. They have properties of both metals and non-metals. Some of the metalloids, such as silicon and germanium, are semi-conductors.
Non-Metals
Groups 14-16 are non-metals. They are not able to conduct heat ore electricity well. They are very brittle, can be a gas (oxygen) or a solid (carbon), have no metallic luster, do not reflect light and have oxidation numbers of +/-4, -3, & -2.
Halogens
Group 17 includes the halogens and are five non-metallic elements. They have 7 electrons in their outer shells and an oxidation number of -1.
Noble Gases
Group 18 are the noble gases. They have an oxidation number of 0. This keeps them from making compounds easily. Also, all noble gases have 8 electrons in their outer shell which makes them very stable.
Rare Earth Elements
There are 30 rare earth elements. They are made up of the lanthanide and actinide series. One of the lanthanide series elements and most of the actinide series elements are synthetic (human-made). These can be found in group 3 of the periodic table, and the 6th and 7th periods.
The Periodic table can be divided into nine families of elements each having similar properties. The families include:
Alkali Metals
Group 1 of the periodic table are the alkali metals. They are highly reactive and do not occur freely in nature. They have just one electron in their outer shell, and they are ready to lose it in ionic bonding with other elements. They are malleable, ductile, are good conductors of heat and electricity and are softer than most metals.
Alkaline Metals
Group 2 includes the alkaline earth elements. They are metallic elements and have an oxidation number of +2, making them very reactive.
The Transition Metals
Groups 3 through 12 include 38 elements called transition metals. They are malleable and ductile and also conduct heat and electricity. They have valence electrons in more than one shell and exhibit several common oxidation states.
Other Metals
Groups 13, 14, & 15 include the other metals elements. They are malleable and ductile but are not the same as transition elements. They do not exhibit a variety of oxidation states, they have valence electrons only in the outer shells. They are solid, have high density, and are opaque. Their oxidation numbers are +3, +/14, and -3.
Metalloids
Metalloids are found between the metals and non-metals along a boundary. They have properties of both metals and non-metals. Some of the metalloids, such as silicon and germanium, are semi-conductors.
Non-Metals
Groups 14-16 are non-metals. They are not able to conduct heat ore electricity well. They are very brittle, can be a gas (oxygen) or a solid (carbon), have no metallic luster, do not reflect light and have oxidation numbers of +/-4, -3, & -2.
Halogens
Group 17 includes the halogens and are five non-metallic elements. They have 7 electrons in their outer shells and an oxidation number of -1.
Noble Gases
Group 18 are the noble gases. They have an oxidation number of 0. This keeps them from making compounds easily. Also, all noble gases have 8 electrons in their outer shell which makes them very stable.
Rare Earth Elements
There are 30 rare earth elements. They are made up of the lanthanide and actinide series. One of the lanthanide series elements and most of the actinide series elements are synthetic (human-made). These can be found in group 3 of the periodic table, and the 6th and 7th periods.
