Structure of an atom
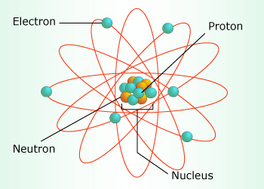
The atom is the basic unit of an element. It can not be broken down chemically. The atom is composed of particles called protons, electrons, and neutrons. It also has a nucleus. The nucleus is made up of protons and electrons with neutrons in the outer shell. Protons have a positive (+) charge, the electrons have no charge (0) and the neutrons have a negative (-) charge.
Let's breakdown the atom! The nucleus is the core of the atom. It was discovered in 1911, but its parts were not identified until 1932. The nucleus is composed of protons and electrons and is held together by a strong force, one of the four basic forces in the world.
The proton is a positively charged particle found within the atomic nuclei. The number of protons in the atom is what define what type of element it is. These numbered protons in the atom are the same as the atomic number of the element. The chemical behavior of the element is also determined by the number of protons.
The electron is a negatively charged particle found within the atom. The electrons are electrically attracted to the positively charged protons and surround the atomic nucleus in pathways called orbitals. The configuration of the orbitals determine the atom's properties.
The neutron is an uncharged particle found within the atomic nuclei. It's mass is slightly larger than the proton.
Let's breakdown the atom! The nucleus is the core of the atom. It was discovered in 1911, but its parts were not identified until 1932. The nucleus is composed of protons and electrons and is held together by a strong force, one of the four basic forces in the world.
The proton is a positively charged particle found within the atomic nuclei. The number of protons in the atom is what define what type of element it is. These numbered protons in the atom are the same as the atomic number of the element. The chemical behavior of the element is also determined by the number of protons.
The electron is a negatively charged particle found within the atom. The electrons are electrically attracted to the positively charged protons and surround the atomic nucleus in pathways called orbitals. The configuration of the orbitals determine the atom's properties.
The neutron is an uncharged particle found within the atomic nuclei. It's mass is slightly larger than the proton.
